Start Slideshow
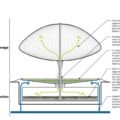 balloon1.jpgWhile the hydrogen fuel cell might not be viable for commercial vehicles for years, here's some hope for the promising element. A group of Philadelphia-based creatives known as the 20/2 Collaborative have designed a unique concept that enables on-site production and distribution of biologically produced hydrogen fuel for vehicles. This plan mixes algae ponds with floating balloons to integrate fuel production and distribution into the local landscape and allows the renewable fuel to be created and distributed from the same place. This may sound a little complicated, but the renderings sure look amazing. Who knew that fuel production could look like so much fun?1
balloon1.jpgWhile the hydrogen fuel cell might not be viable for commercial vehicles for years, here's some hope for the promising element. A group of Philadelphia-based creatives known as the 20/2 Collaborative have designed a unique concept that enables on-site production and distribution of biologically produced hydrogen fuel for vehicles. This plan mixes algae ponds with floating balloons to integrate fuel production and distribution into the local landscape and allows the renewable fuel to be created and distributed from the same place. This may sound a little complicated, but the renderings sure look amazing. Who knew that fuel production could look like so much fun?1 balloon2.jpgWhile the hydrogen fuel cell might not be viable for commercial vehicles for years, here's some hope for the promising element. A group of Philadelphia-based creatives known as the 20/2 Collaborative have designed a unique concept that enables on-site production and distribution of biologically produced hydrogen fuel for vehicles. This plan mixes algae ponds with floating balloons to integrate fuel production and distribution into the local landscape and allows the renewable fuel to be created and distributed from the same place. This may sound a little complicated, but the renderings sure look amazing. Who knew that fuel production could look like so much fun?2
balloon2.jpgWhile the hydrogen fuel cell might not be viable for commercial vehicles for years, here's some hope for the promising element. A group of Philadelphia-based creatives known as the 20/2 Collaborative have designed a unique concept that enables on-site production and distribution of biologically produced hydrogen fuel for vehicles. This plan mixes algae ponds with floating balloons to integrate fuel production and distribution into the local landscape and allows the renewable fuel to be created and distributed from the same place. This may sound a little complicated, but the renderings sure look amazing. Who knew that fuel production could look like so much fun?2 balloon3.jpgWhile the hydrogen fuel cell might not be viable for commercial vehicles for years, here's some hope for the promising element. A group of Philadelphia-based creatives known as the 20/2 Collaborative have designed a unique concept that enables on-site production and distribution of biologically produced hydrogen fuel for vehicles. This plan mixes algae ponds with floating balloons to integrate fuel production and distribution into the local landscape and allows the renewable fuel to be created and distributed from the same place. This may sound a little complicated, but the renderings sure look amazing. Who knew that fuel production could look like so much fun?3
balloon3.jpgWhile the hydrogen fuel cell might not be viable for commercial vehicles for years, here's some hope for the promising element. A group of Philadelphia-based creatives known as the 20/2 Collaborative have designed a unique concept that enables on-site production and distribution of biologically produced hydrogen fuel for vehicles. This plan mixes algae ponds with floating balloons to integrate fuel production and distribution into the local landscape and allows the renewable fuel to be created and distributed from the same place. This may sound a little complicated, but the renderings sure look amazing. Who knew that fuel production could look like so much fun?3 balloon4.jpgWhile the hydrogen fuel cell might not be viable for commercial vehicles for years, here's some hope for the promising element. A group of Philadelphia-based creatives known as the 20/2 Collaborative have designed a unique concept that enables on-site production and distribution of biologically produced hydrogen fuel for vehicles. This plan mixes algae ponds with floating balloons to integrate fuel production and distribution into the local landscape and allows the renewable fuel to be created and distributed from the same place. This may sound a little complicated, but the renderings sure look amazing. Who knew that fuel production could look like so much fun?4
balloon4.jpgWhile the hydrogen fuel cell might not be viable for commercial vehicles for years, here's some hope for the promising element. A group of Philadelphia-based creatives known as the 20/2 Collaborative have designed a unique concept that enables on-site production and distribution of biologically produced hydrogen fuel for vehicles. This plan mixes algae ponds with floating balloons to integrate fuel production and distribution into the local landscape and allows the renewable fuel to be created and distributed from the same place. This may sound a little complicated, but the renderings sure look amazing. Who knew that fuel production could look like so much fun?4 balloon5.jpgWhile the hydrogen fuel cell might not be viable for commercial vehicles for years, here's some hope for the promising element. A group of Philadelphia-based creatives known as the 20/2 Collaborative have designed a unique concept that enables on-site production and distribution of biologically produced hydrogen fuel for vehicles. This plan mixes algae ponds with floating balloons to integrate fuel production and distribution into the local landscape and allows the renewable fuel to be created and distributed from the same place. This may sound a little complicated, but the renderings sure look amazing. Who knew that fuel production could look like so much fun?5
balloon5.jpgWhile the hydrogen fuel cell might not be viable for commercial vehicles for years, here's some hope for the promising element. A group of Philadelphia-based creatives known as the 20/2 Collaborative have designed a unique concept that enables on-site production and distribution of biologically produced hydrogen fuel for vehicles. This plan mixes algae ponds with floating balloons to integrate fuel production and distribution into the local landscape and allows the renewable fuel to be created and distributed from the same place. This may sound a little complicated, but the renderings sure look amazing. Who knew that fuel production could look like so much fun?5 balloon6.jpgWhile the hydrogen fuel cell might not be viable for commercial vehicles for years, here's some hope for the promising element. A group of Philadelphia-based creatives known as the 20/2 Collaborative have designed a unique concept that enables on-site production and distribution of biologically produced hydrogen fuel for vehicles. This plan mixes algae ponds with floating balloons to integrate fuel production and distribution into the local landscape and allows the renewable fuel to be created and distributed from the same place. This may sound a little complicated, but the renderings sure look amazing. Who knew that fuel production could look like so much fun?6
balloon6.jpgWhile the hydrogen fuel cell might not be viable for commercial vehicles for years, here's some hope for the promising element. A group of Philadelphia-based creatives known as the 20/2 Collaborative have designed a unique concept that enables on-site production and distribution of biologically produced hydrogen fuel for vehicles. This plan mixes algae ponds with floating balloons to integrate fuel production and distribution into the local landscape and allows the renewable fuel to be created and distributed from the same place. This may sound a little complicated, but the renderings sure look amazing. Who knew that fuel production could look like so much fun?6







