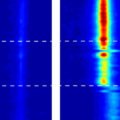
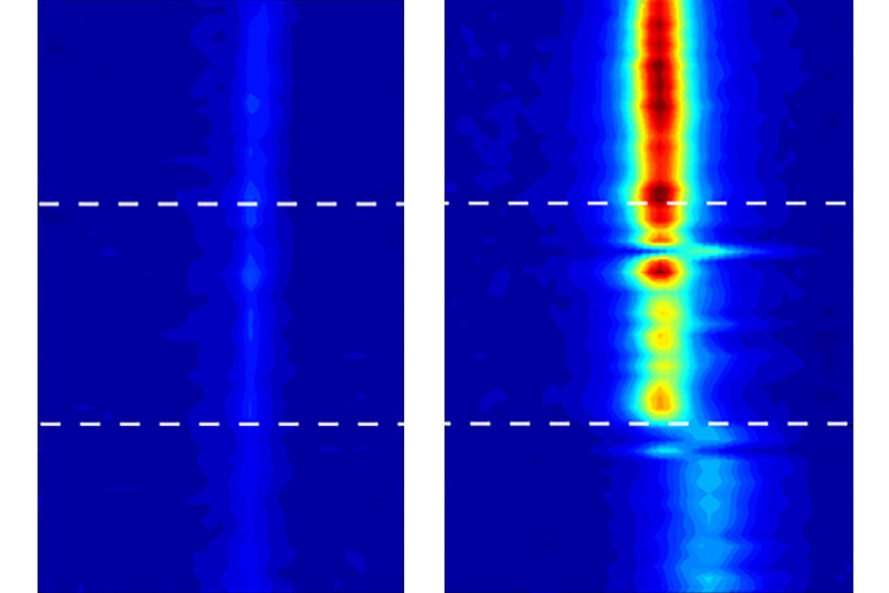
When water, in its liquid form, is confined within carbon nanotubes, it takes on some amazing new properties. Researchers at MIT have discovered that water trapped inside carbon nanotubes can actually freeze at the high temperatures that would normally bring it to a rolling boil. Previous research has long shown that the boiling and freezing points of water change when it is confined to small spaces, but those temperature variations usually hover around 10C. The introduction of carbon nanotubes has changed the game significantly.
Carbon nanotubes are tube-like structures with a diameter measured in nanometers, which are equal to one-billionth of a meter or about 10,000 times smaller than a human hair. The carbon nanotubes used during the MIT experiments were just slightly larger in diameter than the width of a few water molecules. Because water confined within the carbon nanotubes can take on a solid frozen state at a much higher temperature than in other vessels, the discovery could lead to inventions such as ice-filled wires, which could exist at room temperature.
Related: MIT uses carbon nanotubes to boost lithium battery power 10x
In order to better understand how water molecules behave when trapped in such small spaces, the research team used carbon nanotubes of different diameters, noting that even a tiny fraction of difference in size translated into different phase change temperature points. Nanotubes ranging from 1.05 nanometers to 1.06 nanometers resulted in a difference of tens of degrees around the apparent freezing point, something that surprised the research team. Michael Strano, the Carbon P. Dubbs Professor in Chemical Engineering at MIT, is one of five contributing authors on the research. “If you confine a fluid to a nanocavity, you can actually distort its phase behavior,” he said. “The effect is much greater than anyone had anticipated.”
The research was recently published in the journal Nature Nanotechnology.
Via New Atlas
Images via Cloudzilla/Flickr and MIT